The DLSU Research Ethics Review Committee (RERC) has been established since January 2017. The RERC is an independent ethics review body composed of representatives of the various colleges of the university.
In light of the increased research productivity in the University, a new ethics governance structure was created in 2022. The review process is now conducted by multidisciplinary research ethics review panels (RERP) under the administration of the University RERC.
The RERC has been tasked to ensure that research of affiliated members of the University are of the highest standards of scientific integrity and ethical accountability. The primary responsibilities of the RERC are identifying and assessing the risks and the potential benefits of a research, as well as providing guidance for adherence to the University Code for the Responsible Conduct of Research, and updated international, national, and disciplinal guidelines on research ethics.
In line with the University’s vision and mission, the RERC, shall ensure that the health, welfare, dignity and rights of human participants are safeguarded, and that risks to participants, researchers, third parties and to the university are minimized, and to protect the integrity of the research and the data.
The DLSU RERC presently holds Level 2 accreditation status conferred by the PHREB. The accrediting body is the Philippine Health Research Ethics Board (PHREB), which is a government agency that was created by virtue of the Philippine National Health Research System Act of 2013 (RA 10352) under the Department of the Science and Technology Special Order No. 091 s. 2006 “to ensure adherence to the universal principles for the protection of human participants in research studies conducted in the Philippines”. Being Level 2 accredited means that the University RERC can review all types of research except clinical trials required for FDA registration of new drugs as these may entail more than minimal risk to participants.
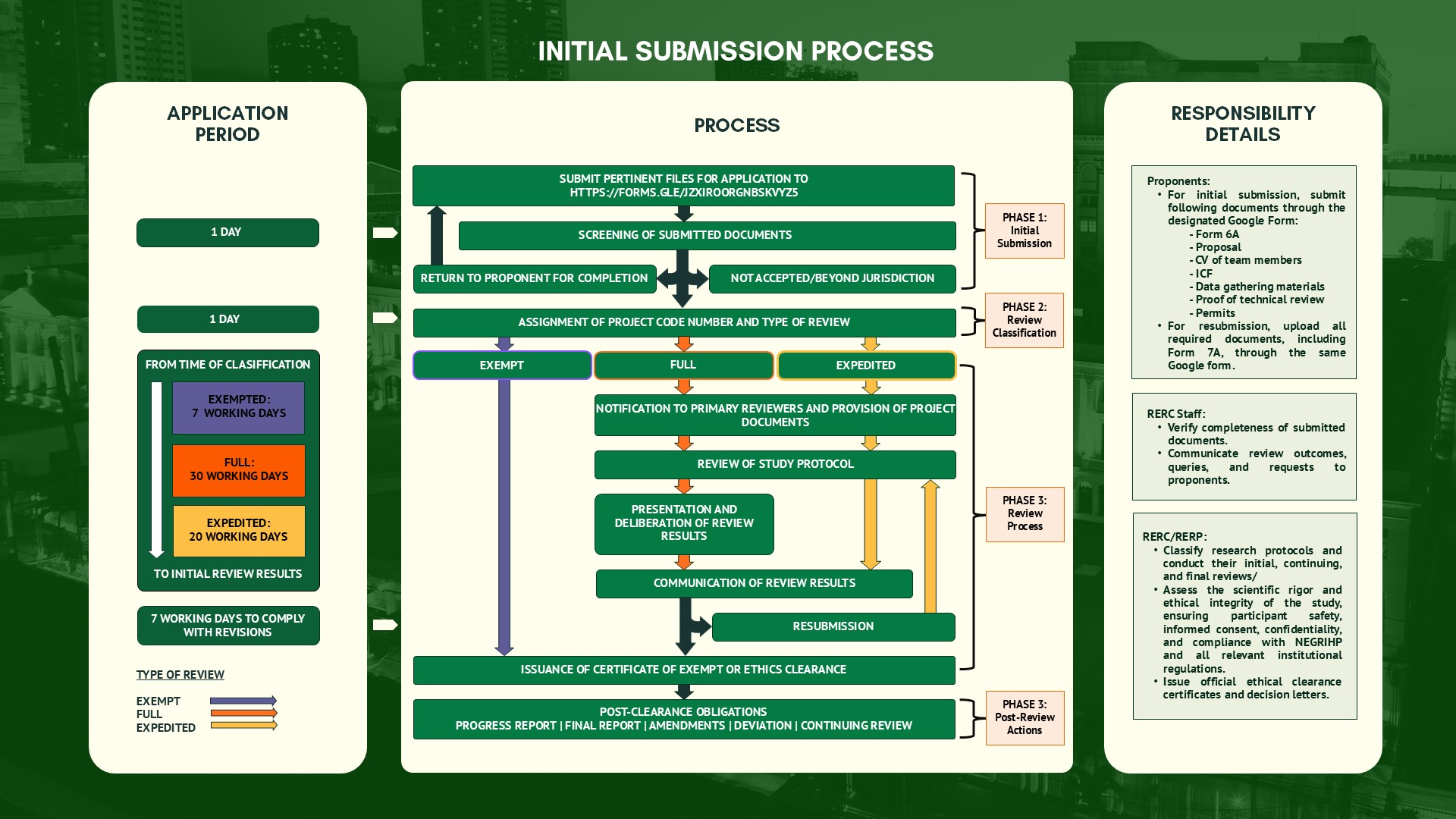
I. Ethics Review Application
- Fill out RERC Form 6A and attach the corresponding files needed. You may download the files here: Application for Ethics Review
- Label your files accordingly:
- Proposal, version #, date submitted: Proposal, version 1, 01 January 2023
- Informed Consent Form, version #, date submitted: Informed Consent Form, version 1, 01 January 2023
- Data Gathering Materials, version #, date submitted, Questionnaire, version 1, 01 January 2023.
- Kindly include also a certificate or approval signed by BOTH the thesis adviser and panel member/s, if your submission is your thesis/course requirement.
- Curriculum vitae of lead, co-researchers, and thesis adviser* must be attached
- Informed consent form and data gathering materials must be attached.
- Submit the files through the Application for Ethics Review Submission Page
*Note: CVs of thesis adviser are required for applications submitted by undergraduate and graduate students
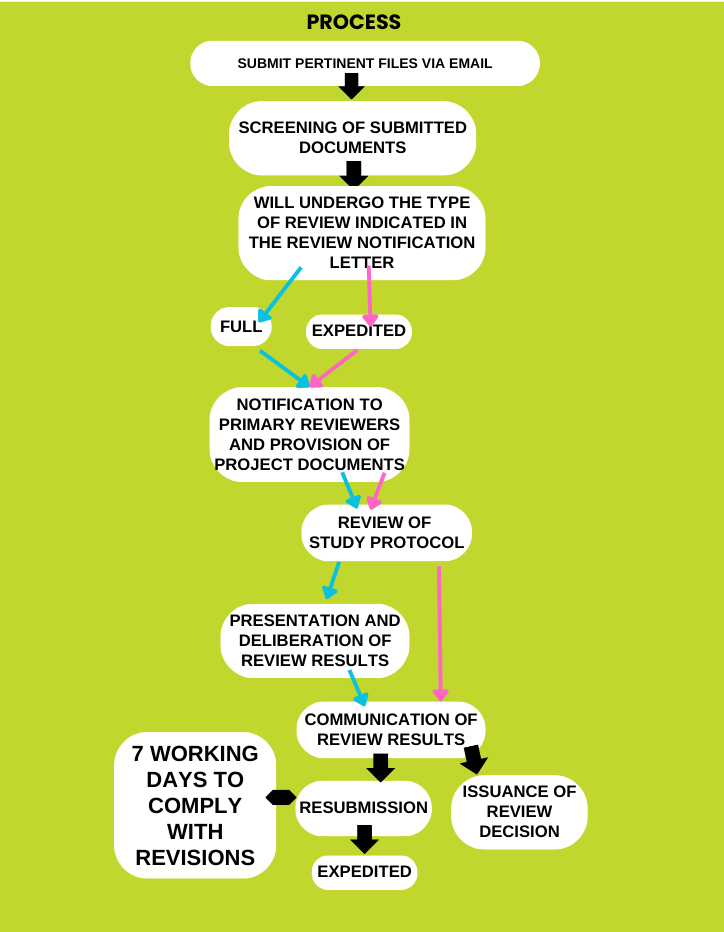
II. Resubmissions
For Resubmissions (i.e., revisions in response to recommendations given by the RERP), kindly indicate it is a resubmission and upload in this Ethics Review Form.
III. Other Submissions
For Progress Report, Reports on RNE/SAE, Reports on Deviations, Requests for Amendments, Application for Continuing Review, and Final Report, fill out appropriate forms (provide link to page of SOP and Forms tab) and send to [email protected]. The email subject should include the project code given after initial review.
Example: 2024-XXX Progress Report Submission
Term 1, AY 2025-2026
(September 1, 2025 – December 6, 2025)
Cut-offs for ethics review applications for Term 1 AY 2025-2026:
| Month | Every Friday |
| August | 29 August 2025 |
| September | 5 September 2025
12 September 2025 19 September 2025 26 September 2025 |
| October | 3 October 2025
10 October 2025 17 October 2025 24 October 2025 |
| November | 7 November 2025
14 November 2025 21 November 2025 28 November 2025 |
Classification Meetings (Every Monday*, 9:30AM)
| Month | Every Monday |
| September | 1 September 2025
8 September 2025 15 September 2025 22 September 2025 29 September 2025 |
| October | 6 October 2025
13 October 2025 20 October 2025 27 October 2025 |
| November | 10 November 2025
17 November 2025 24 November 2025 |
| December | 1 December 2025 |
Research Ethics Review Panel Meetings (Every Wednesday)
Note: dates are subject to change
| Month | Panel 1 | Panel 2 | Panel 3 | Panel 4 |
| September | 3 September 2025 | 10 September 2025 | 17 September 2025 | 24 September 2025 |
| October | 1 October 2025 | 8 October 2025 | 15 October 2025 | 22 October 2025 |
| November | 5 November 2025 | 12 November 2025 | 19 November 2025 | 26 November 2025 |
Research Ethics Review Committee Meetings
(2nd Tuesday of the Month, Classification at 3:00PM* then RERC Meeting)
| Month | Date |
| September | 9 September 2025* |
| October | 7 October 2025 |
| November | 11 November 2025 |
| December | 9/10 December 2025* |
Selection and Appointment of Members
SOP 1A: Selection and Appointment of Research Ethics Committee Members
- RERC Form 1A: Nominee Curriculum Vitae
- RERC Form 1B: RERC Member Terms of Reference
- RERC Form 1C: Confidentiality Agreement
- RERC Form 1D: Conflict of Interest Declaration
SOP 1B: Selection and Appointment of Research Ethics Review Panel Members
- RERC Form 1A: Nominee Curriculum Vitae
- RERC Form 1B: RERC Member Terms of Reference
- RERC Form 1C: Confidentiality Agreement
- RERC Form 1D: Conflict of Interest Declaration
SOP 1C: Formation of Research Ethics Review Panels
SOP 2: Designation of RERC Officers
SOP 3: Appointment of Consultants
- RERC Form 3A: Invitation Letter for Independent Consultant
- RERC Form 3B: Appointment Letter for Independent Consultant
- RERC Form 3C: Terms of Reference for Independent Consultant
- RERC Form 1C: Confidentiality Agreement
- RERC Form 1D: Conflict of Interest Declaration
Types of Review
- RERC Form 4A: Research Project Assessment Form
- RERC Form 4B: Informed Consent Assessment Form
- RERC Form 20A: Notice of Review Results-Approved
- RERC Form 20B: Notice of Review Results-Modifications Required
- RERC Form 4F: Review Results
- RERC Form 5A: Notification for Full Review
- RERC Form 4A: Project Assessment Form
- RERC Form 4B: Informed Consent Assessment Form
- RERC Form 20A: Notice of Review Results-Approved
- RERC Form 20B: Notice of Review Results-Modifications Required
- RERC Form 5B: Full Review Report Form
- RERC From 5C: Full Review Decision Guide
Management of Submissions
SOP 6: Management of Initial Submissions (Classification)
- RERC Form 6A: Application for Ethics Review
- RERC Form 6B: Classification of Review Form
- RERC Form 6C: Guidelines for Review Classification
- RERC Form 6D: Certificate of Exemption
- RERC Form 6E: Exemption Checklist
- RERC Form 6F: Notice of Expedited Review
- RERC Form 6G: Notice of Full Review
- RERC Form 6H: Endorsement Letter Template
SOP 7: Management of Resubmissions
- RERC Form 7A: Cover Letter for Resubmissions
- RERC Form 7B: Review of Resubmissions
- RERC Form 4F: Review Results
- RERC Form 20A: Notice of Review Results – Approved
- RERC Form 20B: Notice of Review Results – Modifications Required
Progress Report
SOP 8 Review of Progress Report
- RERC Form 8A: Progress Report
- RERC Form 8B: Progress Report Decision Letter
- RERC Form 8C: Progress Report Assessment Form
Amendments
- RERC Form 9A: Amendment Request Form
- RERC Form 9B: Amendment Request Decision Letter
- RERC Form 9C: Amendment Request Assessment Form
Management of Research Deviation and Violation
SOP 10 Management of Research Deviation and Violation Report
- RERC Form 10A: Research Project Deviation/ Violation Report Form
- RERC Form 10B: Project Deviation Decision Letter
Safety Report
Continuing Review
SOP 12 Management of an Application for Continuing Review
- RERC Form 12A: Application for Continuing Review
- RERC Form 12B: Continuing Review Decision Letter
- RERC Form 12C: Continuing Review Results
Final Report
- RERC Form 13A: Final Report Form
- RERC Form 13B: Final Report Decision Letter
- RERC Form 13C: Final Report Assessment Form
- RERC Form 4F: Review Results
Review of Early Termination
SOP 14 Review of Early Termination Reports
- RERC Form 14A: Early Termination Report
- RERC Form 14B: Early Termination Decision Letter
- RERC Form 14C: Early Termination Assessment Form
- RERC Form 4F: Review Results
Management of Appeals, Complaints, and Queries
Management and Preparation of Meetings
Management of Communication
SOP 20 Communication on RERC and RERP Decisions
- RERC Form 20A: Notice of Review Results-Approved
- RERC Form 20B: Notice of Review Results-Modifications Required
- RERC Form 4F: Review Results
- RERC Form 5A: Notification Letter-Full Review
- RERC Form 5E: Full Review Report Form
- RERC Form 8B: Progress Report Decision Letter
- RERC Form 9B: Amendment Request Decision Letter
- RERC Form 10B: Project Deviation Decision Letter
- RERC Form 11C: RNE/ SAE Decision Letter
- RERC Form 12B: Continuing Review Decision Letter
- RERC Form 13B: Final Report Decision Letter
- RERC Form 14B: Early Termination Decision Letter
- RERC Form 15A: Decision Letter for Appeals
Management of Files
Writing and Revision of SOP
Appendix
Clearance Validity
| Start Date | End Date |
Research Ethics Review Committee (RERC) Members
| Position | Name |
| RERC Chair | Ms. Nathalie Rose Lim-Cheng |
| RERC Vice-Chair | Dr. Myla Arcinas |
| RERC Secretary | Dr. Michelle Natividad |
| RERC Member | Dr. Aileen Bautista-Del Rosario |
| RERC Member | Dr. Wilfred Espulgar |
Research Ethics Review Panel (RERP) Members
| Albao, Marycris | Javier, Ma. Elena | Pakingan, Karmia |
| Baquillas, Jonalyn | Go, Stella | Rodriguez, Cristina |
| Dominguez, Maribel | Jimenez, Shieradel | Chua, Nellie Margareth |
| Co, Homer | Ranieses, Jessica | Simbillo, Janus Aries |
| Duay, Searle Aichelle | Manlagnit, Alicia | Sumayao Jr., Rodolfo |
| Ello, Aileen | Marcos, Sernando | Tanhueco, Renan |
| Gan, Rosita | Melegrito, Ma. Lourdes | Torres, Jose Victor |
| Garcia, Joel | Tiglao-Torres, Amaryllis | Gastardo-Conaco, Maria Cecilia |
| Natividad, Filipinas | Valdez, Eduardo Victor | Zapanta, Reinier Dave |
| Martin, Christian | Alpay, Judith |
We kindly request to be contacted through email for us to have a clear record of our correspondence.
For inquiries, follow ups, or submissions (after initial review), please email [email protected]. Unless it is a general inquiry, the email subject should indicate the RERC-assigned project code.
For serious concerns (e.g., reporting complaints, violations, or non-compliance to ethical practices), please email [email protected].